Introduction
Herbicides control weeds by interfering with how they grow. This is achieved by a number of different ‘modes of action’ (MOA) which all either ultimately stop seeds from germinating or establishing as seedlings; prevent plants from making essential carbohydrates, proteins or lipids (oils and fats); or desiccate leaves and stems. Knowing a herbicide MOA is important to understanding how to use a herbicide most effectively. It is a major factor in both herbicide selectivity and weed resistance.
 Phytotoxicity symptoms can indicate mode of action
The symptoms observed on weeds sprayed with herbicides express the MOA. In herbicide R&D, when new chemicals are screened experts carefully observe the detail and timing of the appearance of symptoms to gain clues as to the MOA. Fully understanding a MOA may take years of research by plant physiologists, biochemists, molecular biologists and many other scientific disciplines. The precise MOA of paraquat is very well understood - for more information, click here.
Phytotoxicity symptoms can indicate mode of action
The symptoms observed on weeds sprayed with herbicides express the MOA. In herbicide R&D, when new chemicals are screened experts carefully observe the detail and timing of the appearance of symptoms to gain clues as to the MOA. Fully understanding a MOA may take years of research by plant physiologists, biochemists, molecular biologists and many other scientific disciplines. The precise MOA of paraquat is very well understood - for more information, click here.
The more that is known about MOA, the more safely and effectively herbicides can be used. There are even examples of pharmaceuticals being developed as a result of research into herbicide MOA . For example, the drug nitisinone has now replaced the need for a liver transplant as the first-line response to type 1 hereditary tyrosinaemia, a rare metabolic disorder in children. Nitisinone was originally being studied as a potential herbicide. Its MOA is the inhibition of the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD), the same as several commercial herbicides including Syngenta’s mesotrione (Callisto)1.
Mode of action in detail
Most often, herbicides have MOAs involving a target enzyme that no longer functions properly, or at all. This is usually because the herbicide molecule has distorted the enzyme molecule in some way. Shape is crucial to enzyme function. Enzymes are ‘catalysts’. They provide a platform for a specific chemical reaction that they are not directly part of and that would not happen effectively without them. ‘Knocking-out’ an enzyme with a herbicide has two main effects with various consequences:
- Component chemicals for the reaction accumulate and may be directly or indirectly damaging.
- Absence of the reaction’s product will restrict growth, either through starvation of key building blocks or because the reaction makes chemicals which normally protect the plant.
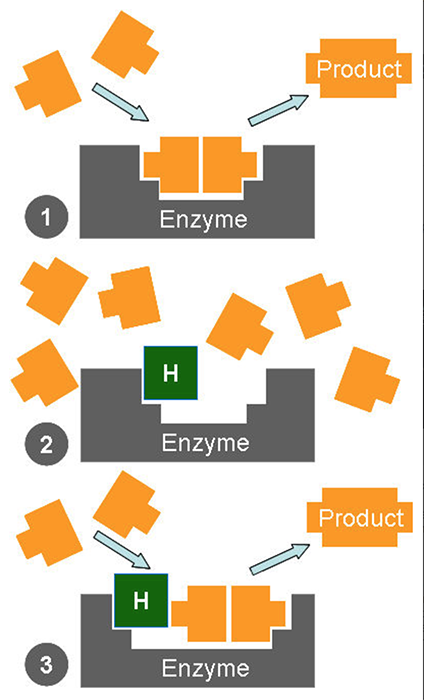 How enzymes work
Diagram 1 shows an enzyme functioning normally. In Diagram 2 a herbicide is stopping biochemicals coming together to form the usual product. As a result the plant is starved of the product and potentially damaging building blocks accumulate. Diagram 3 shows an enzyme which has mutated so that even when the herbicide is present the reaction can continue. Either the herbicide is selective to species with this type of enzyme, or an individual plant with this mutation could reproduce to establish a population of resistant weeds.
How enzymes work
Diagram 1 shows an enzyme functioning normally. In Diagram 2 a herbicide is stopping biochemicals coming together to form the usual product. As a result the plant is starved of the product and potentially damaging building blocks accumulate. Diagram 3 shows an enzyme which has mutated so that even when the herbicide is present the reaction can continue. Either the herbicide is selective to species with this type of enzyme, or an individual plant with this mutation could reproduce to establish a population of resistant weeds.
As plants have evolved, slight differences in their biochemical systems for carrying-out physiological processes have arisen. Sometimes a gene with DNA coding for a particular enzyme or other herbicide target will have mutated in some species so that variations in that enzyme exist, which differ by maybe only one or two amino acids out of thousands. This small change will still allow plants to grow successfully, but it may mean that herbicides with a MOA involving this enzyme will only be effective on one type and, therefore, on a particular group of species.
Gene mutations are responsible for one sort of herbicide selectivity based on MOA. Herbicides such as fluazifop-p-butyl inhibit the enzyme Acetyl Coenzyme A Carboxylase (ACC-ase) and selectively control grass weeds in broad-leaved crops like soybeans or oilseed rape (canola). The enzyme carrying out the same function in broadleaved weeds and crops is slightly different and not affected by the herbicide. On the other hand, glyphosate is a non-selective herbicide with a very wide weed control spectrum because it inhibits the enzyme 5-enolpyruvylshikimate-3-phosphate (EPSP) synthase, a fundamentally important enzyme very similar in all plants.
Mutations are also one way in which weed resistance to herbicides can occur. Rare individual plants which have a mutated enzyme may not be susceptible to a herbicide which is generally effective on that species. An outbreak of weed resistance occurs when such an individual is allowed to reproduce and comes to dominate the weed population in a field. Ensuring that herbicides with different MOAs are used helps to avoid weeds becoming resistant.
The other main type of selectivity and weed resistance occurs when one group of plants can metabolise a herbicide rapidly enough to avoid any damage.
Several other types of herbicide MOA exist. Some affect photosynthesis in various ways to ultimately divert the flow of energy from sunlight. Instead of being incorporated into chemical energy stores it ends up in very destructive ‘free radicals’ based on oxygen. These destroy membranes and desiccate leaves. Another large group, the first selective herbicides to be discovered, mimic plant hormones – the chemical messengers that control growth and development. Another group affect cell division by, for instance, disrupting the mechanism by which chromosomes are arranged in a cell as it prepares to divide in two. Others herbicides have more general chemical effects which are hard describe as specific MOAs, or where very little has been discovered about the MOA. These are generally older products, which have been superseded.
Classifying modes of action
Herbicide MOAs can be classified by the plant process affected, e.g. photosynthesis, cell division or specific enzyme targets. This often reflects the practical uses of herbicide products. For instance, those affecting photosynthesis are applied post-emergence and are often fast acting; those affecting cell division are often pre-emergence herbicides affecting germinating seeds; and some enzyme targets will determine patterns of weed control spectrum and selectivity depending on whether the enzyme is fundamental to the growth of all plants or whether it is confined to, say, grasses or broadleaved weeds.
However, there are always significant exceptions to these general rules and their particular chemistry may mean that they move in plants and soil differently. These factors determine selectivity, whether they are contact or systemic herbicides, and whether they can be used pre-emergence, post-emergence, or both.
A more detailed classification is required for managing weed resistance and some web resources are listed at the end of this article.
1. MOAs involving photosynthesis
Four types of herbicides primarily affect photosynthesis. Paraquat and its sister herbicide diquat are the only members of the first type. When the energy in sunlight is captured by chlorophyll it is transferred in a flow of electrons through ‘Photosystem I’. Paraquat diverts this flow resulting in the production of highly reactive free radicals which very quickly destroy cell membranes, spilling the contents, and appearing as yellowing and desiccation. This happens within hours in bright sunlight because of the high levels of energy running rampant. Almost all green plants are affected by paraquat making it a broad-spectrum, non-selective herbicide.
The second type blocks the transfer of energy through ‘Photosystem II’. Herbicides of this type (‘PS II inhibitors’) bind to a protein involved in the transfer chain, reducing its effectiveness. This causes some diversion of electrons with similar, but slower, results to paraquat and slows growth because of less energy for photosynthesis. Triazines like atrazine and ureas like chlortoluron are both PSII inhibitors, but have slightly different MOAs because they bind to the protein concerned in different places.
A third type, the protoporphorinogen oxidase (PPO) inhibitors, interfere with an enzyme involved in producing chlorophyll and other large molecules important in photosynthesis. Without new chlorophyll leaves yellow and photosynthesis slows. However, unused building blocks of chlorophyll accumulate and react with oxygen to form reactive free radicals which are especially destructive to broadleaved species.
The fourth type of photosynthesis inhibitor prevents the production of leaf pigments called carotenoids. These are familiar as the colours of autumn when green chlorophyll has degraded. Carotenoids have roles in protecting chlorophyll from being destroyed by more light energy than it can process. Herbicides in this category have several different targets, but all result in treated weeds being bleached white.
For simplicity, a fifth type can be added. This has a single commercial member, glufosinate. Although it is an enzyme inhibitor, it does indirectly affect photosynthesis and its faster appearing symptoms are due to membrane destruction resulting in desiccation.
| MOA |
Effect on Weeds |
|---|---|
|
Photosystem I (PSI) inhibition |
|
|
Photosystem II (PSII) inhibition |
|
|
Protoporphyrinogen oxygenase (PPO) inhibition |
|
|
Carotenoid biosynthesis inhibition, e.g. 4-hydroxyphenyl-pyruvate-dioxygenase (HPPD) |
|
|
Glutamine synthetase |
|
2. MOAs targeting other enzymes
In this group, the primary effects are to starve weeds of the essential products normally made by the enzymes affected. The appearance of symptoms is usually slow and depends on how conducive the environment is for plant growth. The ACC-ase inhibitors particularly affect growing points at shoot tips, while the ALS inhibitors and glyphosate have more general effects due to the ubiquity of the amino acids they deplete.
| MOA |
Effect on Weeds |
|---|---|
|
Acetyl CoA carboxylase (ACC-ase) inhibition |
|
|
Acetolactate synthase (ALS) inhibition |
|
|
EPSP synthase |
|
3. Other MOAs
Generally less is known about the detail of these other MOAs.
| MOA |
Effect on Weeds |
|---|---|
|
Cell division disruption |
|
|
Seedling growth inhibitors |
|
|
Synthetic auxins |
|
Conclusions
 Frequency of introduction of new herbicide MOAs (refs 2, 3, 4)
Understanding herbicide MOA is important in both R&D and in practical use. New products are continuously launched, but a new MOA is increasingly rare. In fact, with the exception of a few minor herbicides whose MOAs have never been discovered, all of the nearly 300 active ingredients ever commercialised can be included in no more than 25 MOAs. It’s salutary to realise that the ‘golden days’ of discovery seem long gone with a sharp tail-off after the early Seventies and the last new MOA was HPPD inhibition.
Frequency of introduction of new herbicide MOAs (refs 2, 3, 4)
Understanding herbicide MOA is important in both R&D and in practical use. New products are continuously launched, but a new MOA is increasingly rare. In fact, with the exception of a few minor herbicides whose MOAs have never been discovered, all of the nearly 300 active ingredients ever commercialised can be included in no more than 25 MOAs. It’s salutary to realise that the ‘golden days’ of discovery seem long gone with a sharp tail-off after the early Seventies and the last new MOA was HPPD inhibition.
Weed resistance to herbicides like glyphosate threatens food production and means the intelligent use of the small range of MOAs available is vitally important.
References
- Santra S & Baumann, U (2008). Experience of nitisinone for the pharmacological treatment of hereditary tyrosinaemia type 1. Expert Opinion on Pharmacotherapy, 9, (7), 1229-1236
- International Survey of Herbicide Resistant Weeds
- Herbicide Resistance Action Committee (HRAC)
- Powles, S B & Preston C (2006). Evolved glyphosate resistance in plants: biochemical and genetic basis of resistance. Weed Technology, 20, (6), 282-289

